Biotechnology: Leading the Search for a Cure for COVID-19
Biotechnology: Leading the Search for a Cure for COVID-19
by Jonas Calsbeek
Developing a test, therapy, or vaccine is an expensive process, and success is not guaranteed. What role does research in biotechnology play in helping to expedite this process in modern times?
What is COVID-19?
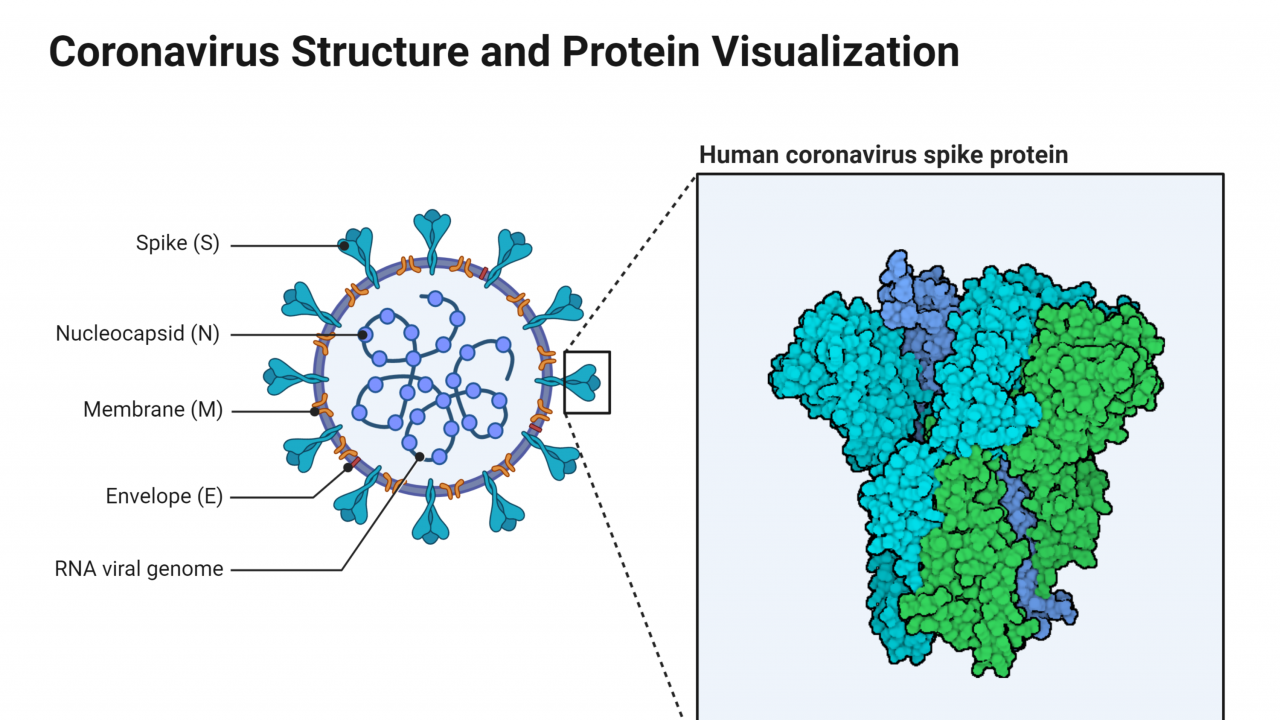
Life as we know it has been turned upside-down in recent months due to the global pandemic caused by COVID-19, the novel coronavirus that emerged late in 2019. Since then, a massive amount of misinformation has been posted online regarding the efficacy of treatments for COVID-19, even the meaning of this acronym has been confused. According to the CDC, the 'CO' stands for 'corona,' 'VI' for 'virus,' and 'D' for disease, and 19 describes the year when the virus became known, 2019.
COVID-19 has shown a remarkable ability to transmit through the human population, with lethal effects, and nothing short of isolation has been successful in stopping its global spread. Since its discovery last year, one of the biggest concerns has been identifying a vaccine to stop this virus. Hospitals around the world are overwhelmed, hundreds of thousands of innocent people are dying, and it is difficult to imagine an end to this healthcare crisis. Fortunately, modern society benefits from a wealth of technology that can be utilized to improve and assist in the search for a cure. Biotechnology plays a key role in the discovery and development of tests, therapies, and vaccines for COVID-19.
Testing for the virus
Testing currently faces a multitude of challenges, including timeliness of results, ability to distribute, and accuracy of the test. When a patient presents to the clinic with symptoms of COVID-19, laboratory tests are available to determine if the virus is present in a mouth or nose swab sample. However, patients who submit these samples often do not hear their results for more than 24 hours due to the time it takes to prepare and run diagnostic tests. Rapid, widespread testing is critical to safely returning society to employment and education endeavors, and this will be difficult with the speed of current technologies. But there is hope! Just this month, research emerged out of UCSF describing the use of CRISPR technology to develop a fast, easy, and accurate COVID-19 test that runs on paper, like an at-home pregnancy test. The researchers confirmed the accuracy of their test using swab samples from COVID-19+ patients in the US. Read more about this new technology in the original article here.
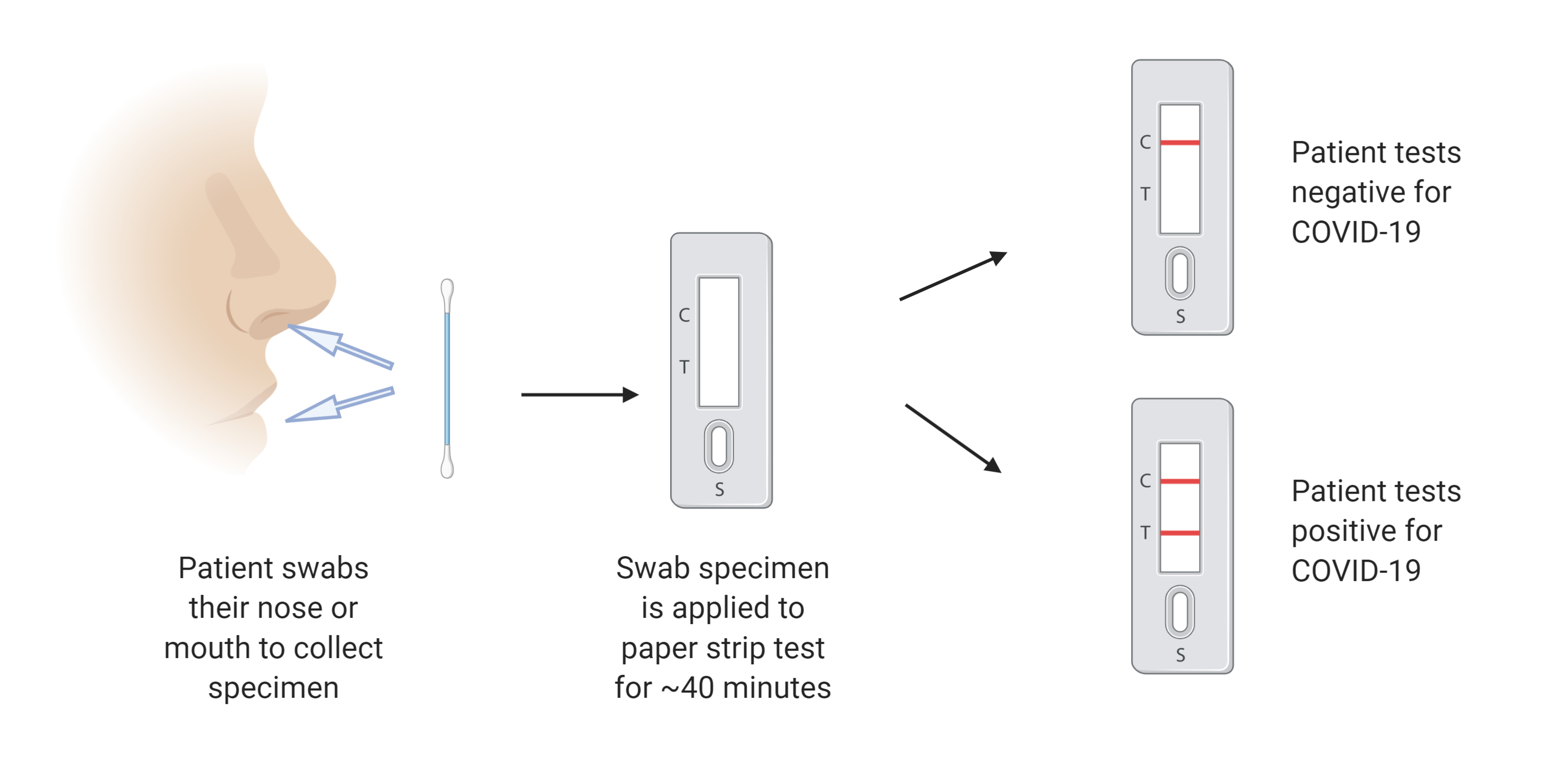 Additional advancements in testing include the recent FDA emergency use authorization for an at-home collection kit made by Labcorp. The first test of its kind to be granted authorization by the FDA, it allows patients at home to collect their own swab samples and mail the kit to LabCorp for processing. While this form of testing still takes some time to get results, its availability helps to increase testing capacity in the USA. While developments in testing are crucial to understanding the spread of the virus, stopping it requires advancement of treatments for patients who test positive.
Additional advancements in testing include the recent FDA emergency use authorization for an at-home collection kit made by Labcorp. The first test of its kind to be granted authorization by the FDA, it allows patients at home to collect their own swab samples and mail the kit to LabCorp for processing. While this form of testing still takes some time to get results, its availability helps to increase testing capacity in the USA. While developments in testing are crucial to understanding the spread of the virus, stopping it requires advancement of treatments for patients who test positive.
Treatments for Infection
Current options for a successful COVID-19 therapy include the use of antiviral drugs that are already approved for humans for similar diseases caused by viruses, among others. A recent review from the University of Texas Southwestern Medical Center discusses what is known about current leading COVID-19 therapies. Read it here. To summarize, no proven effective therapy has been found (yet) for COVID-19. Even though several drugs have demonstrated efficacy against the novel coronavirus when they are applied directly to viral samples, researchers caution against being too optimistic. Preclinical evidence to date relies on experiments that test these potential drugs against the virus in a dish, or in animal models of the disease. While efficacy in these models may predict success in the human, many drugs fail to translate into successes in the clinic due to the complexity of the human body. In the coming months, controlled, randomized clinical trials will determine if any of these drugs will be a lead therapy for human patients suffering from COVID-19. These trials include control groups of patients who do not receive the trial drug, so researchers can determine if a beneficial outcome is a direct result of the drug. Clinical proof that these drugs can stop the virus from infecting the human body must be established before people risk taking a drug that may be toxic.
Alternative Therapeutic Approaches
Other non-pharmacological methods of treating COVID-19 have been recently employed in hospitals, with some success, due to alleviating the symptoms of the disease using ingenuity. One interesting strategy for treating COVID-19 uses blood plasma from patients who have already recovered. Since the surviving patient successfully defeated the virus, circulating immune cells and antibodies can be transferred into a newly infected patient to help them fight the virus, too. The FDA has supported this approach, along with a coalition of hospitals and universities that began the National COVID-19 Convalescent Plasma Project to identify recovered patients willing to donate plasma.
A lot of attention has been drawn to ventilators recently due to the excessive need for these devices to help deliver oxygen to a patient suffering from respiratory disease. A critical limitation of the use of ventilators for COVID-19 symptoms is the limited ability of an infected patient’s lungs to absorb the provided oxygen into the bloodstream. One hallmark of COVID-19 is a respiratory disease like pneumonia, which can make it difficult to absorb enough oxygen through the lung. Clinicians have recently experimented with an invasive process called extracorporeal membrane oxygenation (ECMO) to treat some patients who are not responding well to these conventional treatments. ECMO circulates a patient’s blood outside of the body and passes it through an artificial lung that removes carbon dioxide and adds oxygen directly to the blood before returning it to the body. The FDA also supports this process, recently issuing this guidance on how to expand these efforts during the COVID-19 pandemic.
Vaccine development efforts
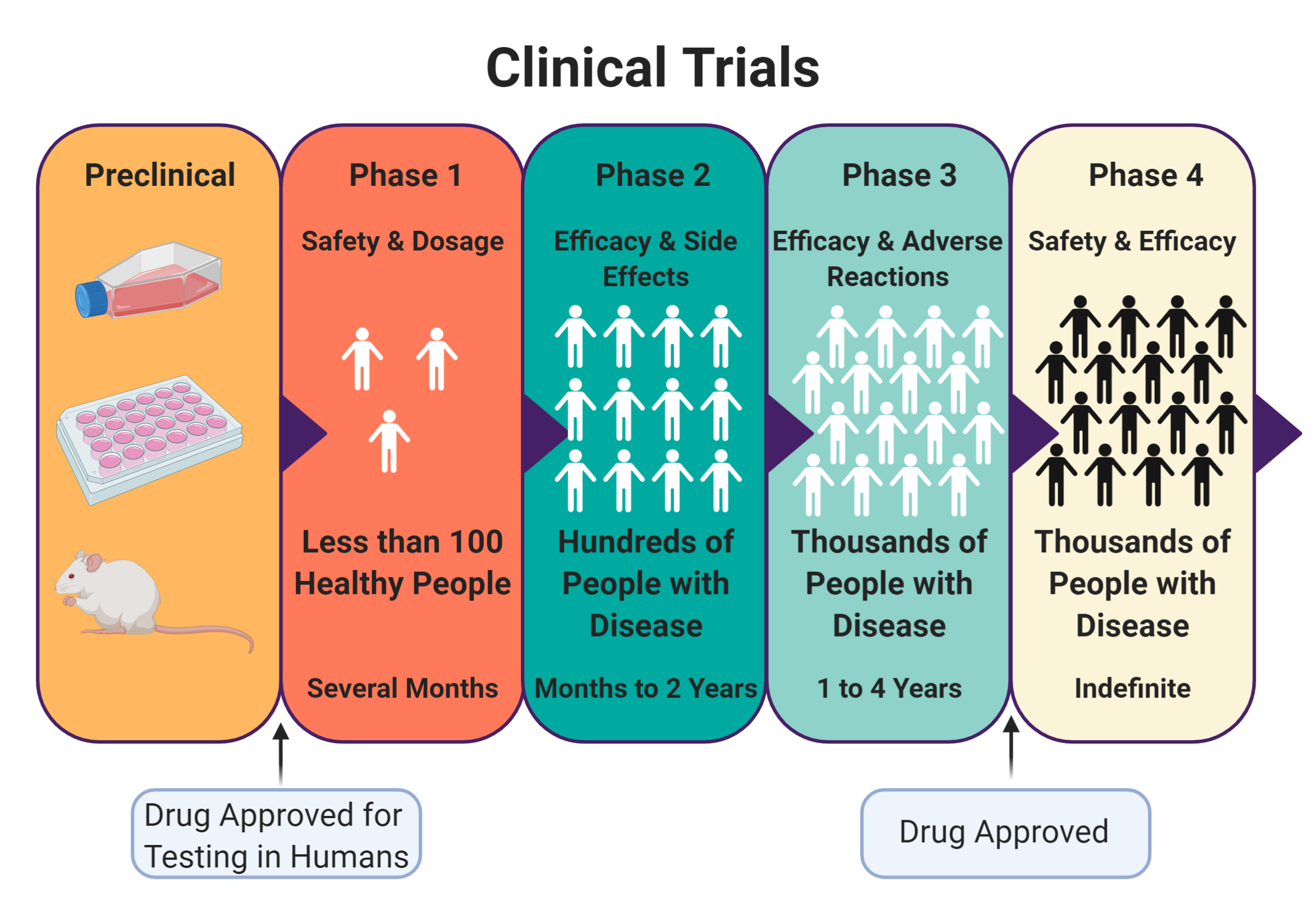 While every hurdle cleared in the race against COVID19 is an achievement, what everyone wants to know about is when a vaccine will be available, but information on that timeline has been as convoluted as the rest of the news regarding this virus. Whether discussing the development of new drugs or new shots, clinical trials are always mentioned in vague terminology that can be confusing for a lay audience. The NIH provides this page to describe why clinical trials are so important to our healthcare system. Preclinical studies in cell or animal models are used to screen entire libraries of compounds to find a few lead candidates likely to be successful in human testing. These leads are submitted for clinical experiments, and the clinical trials are then broken into 4 steps, called phases. In Phase 1 trials, safety is the main concern. Does the vaccine candidate cause any adverse health effects in a small group of healthy volunteers, and what is the right dose? In Phase 2 trials, efficacy is the primary concern. Does the vaccine work to successfully stop the infection in hundreds of volunteers at the dose determined? Phase 3 trials are the final test before approval, where researchers examine diverse populations of patients receiving the candidate vaccine and look for drug-drug interactions or side effects. Finally, Phase 4 trials are designed to run after a vaccine has been approved, because some side effects are not seen until many people have been given a vaccine. Researchers will look back on historical data to determine if any adverse side effects have been noted following approval.
While every hurdle cleared in the race against COVID19 is an achievement, what everyone wants to know about is when a vaccine will be available, but information on that timeline has been as convoluted as the rest of the news regarding this virus. Whether discussing the development of new drugs or new shots, clinical trials are always mentioned in vague terminology that can be confusing for a lay audience. The NIH provides this page to describe why clinical trials are so important to our healthcare system. Preclinical studies in cell or animal models are used to screen entire libraries of compounds to find a few lead candidates likely to be successful in human testing. These leads are submitted for clinical experiments, and the clinical trials are then broken into 4 steps, called phases. In Phase 1 trials, safety is the main concern. Does the vaccine candidate cause any adverse health effects in a small group of healthy volunteers, and what is the right dose? In Phase 2 trials, efficacy is the primary concern. Does the vaccine work to successfully stop the infection in hundreds of volunteers at the dose determined? Phase 3 trials are the final test before approval, where researchers examine diverse populations of patients receiving the candidate vaccine and look for drug-drug interactions or side effects. Finally, Phase 4 trials are designed to run after a vaccine has been approved, because some side effects are not seen until many people have been given a vaccine. Researchers will look back on historical data to determine if any adverse side effects have been noted following approval.
Speeding Up the Search
One challenge in trying to develop a vaccine for an emerging pandemic is that the likelihood of success is less than 10%. For this reason, it is necessary to immediately begin the development of several different vaccines at one time, even if it will waste billions of dollars. If vaccines for COVID-19 are only pursued one at a time, it could be decades before a successful one is scaled to meet the demand to make human populations immune. The Bill and Melinda Gates Foundation is funding the creation of factories to pursue 7 of the lead vaccine candidates, despite the guaranteed loss of money that will result from failures. Mr. Gates said, "Even though we'll end up picking at most two of them, we're going to fund factories for all seven, just so that we don't waste time in serially saying, 'OK, which vaccine works?' and then building the factory." Read more about these efforts here.
Researchers in biomedical research are scrambling to repurpose or develop new technology that can aid in the search, and biotechnology is positioned to be a critical weapon in the fight. One excellent example of this includes researchers from Folding@Home, based in the Washington University in St. Louis School of Medicine. Just this year, they accidentally created the fastest supercomputer on the planet when searching for COVID-19 protein structures. Their site allows users to volunteer their computer hardware to be utilized to simulate protein folding, which can help identify druggable sites or targets in a virus. Because so many people signed up during the pandemic quarantine, their processing power exceeded all the current records set by supercomputers to date. Harnessing technology to expedite discovering a cure is critical to moving humans into a new generation of healthcare, where we can be more proactive than reactive, and be better prepared for the next virus.
Conclusion
Biotechnology research is on the front lines of identifying reliable tests, effective therapies, and scalable vaccines for the virus that causes COVID-19. Testing is beginning to ramp up in the USA, and ongoing scientific efforts to identify therapies for those who are sick are making progress. The major breakthrough in this healthcare crisis will come when there is success in identifying a vaccine to protect humans from COVID-19. Since so much time goes into clinical trials for successful candidates, a lot of money will be wasted on the unsuccessful attempts. However, the hope is that at least one of the vaccines currently in development will prove effective and will be manufactured and scaled quickly to meet the global demand to stop this disease. Until then, life as we know it will change to meet the demands of social distancing requirements to slow the spread of COVID-19.
Images in this post have been created with BioRender.com.
